
Chromatography is a technique used to separate components in a mixture and can be used to partially identify the components. Sepn of Three Component Mixture 1 SEPARATION OF A THREE COMPONENT MIXTURE.
Fitzpatrick Department of Chemistry University of Southwestern Louisiana Lafayette LA 70504-4370 Revised 2003.
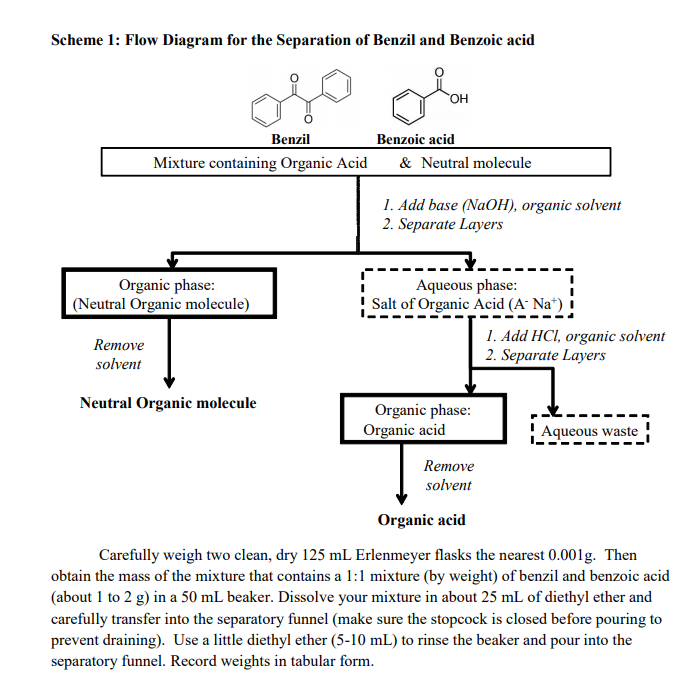
Separation of a three component mixture by extraction lab report. Pre Lab Report 5 - Separation of a three component mixture via Acid Base extraction. Three Component Mixture Separation Introduction In lab this week we will be separating a mixture with three different components acidic basic and neutral. We will separate them using a technique known as acidbase extraction which is a specific type of liquidliquid extraction.
AcidBase extraction is a type of liquidliquid. Pre-Lab_ Separation of a Three Component Mixture Using Extraction Techniquesdocx. Separation of a Three-Component Mixture via AcidBase Extraction Introduction.
During this experiment the purpose is to use certain techniques to separate a three component mixture containing benzoic acid fluorenone and benzocaine. Purpose Statement The purpose of this experiment is to demonstrate the separation of three components that are combined in a mixture that will extract the aqueous layer from the organic layer using different extraction techniques. Introduction The term extraction references the method that is used by organic chemists for the fast unpurified.
Sepn of Three Component Mixture 1 SEPARATION OF A THREE COMPONENT MIXTURE. EXTRACTION EXPERIMENTAL PROCEDURE PROVIDED BY Dr. WAGHULDE Text p 127-140 337-338 Extraction involves transferring a solute substance being dissolved from one solvent liquid used to dissolve the solid into another based on solubility.
The two solvents used. Mixture consisting of an acidic basic and neutral component. In order to separate these three.
Components in a sufficient manner a specific type of liquid-liquid extraction will be performed. Liquid-liquid extraction is the separation of compounds based on their relative solubilities in two. Isolating the Components of a Three-Component Mixture 22712 Lindsay Valentine Partners.
Lauren Flockton Aurora Vellante Objective. The purpose of this experiment is to separate the components of a mixture of sand sodium chloride and calcium carbonate. After the separation the percent by mass of each component in the original mixture and the.
Separation of Components Separation of The Components In A Mixture Lab Report 3 Christian Price Anna Benitez Dr. Ridlen Chemistry 1411 Lab Day. MTW 800PM 17 June 2019 Page 1 Separation of Components Objectives- In this experiment the purpose of this lab is to separate three components of a mixture which includes NaCl sodium chloride NH4Cl ammonium chloride and.
Separation of the Components of a mixture This lab report will discuss the methods of separating substances from one another using decantation extraction and sublimation techniques. The mixture that is being used in this experiment is a mixture of sodium chloride NaCl ammonium chloride NH 4 Cl and silicon dioxide SiO 2. CHEM 3401AB March 7 2019 TA.
Maya Handberry Separation by Extraction Purpose Extraction is a separation technique used to isolate different components from a mixture. 1 When performing an extraction experiment the use of two immiscible compounds is crucial because it allows for the separation of mixture into two layers. An organic layer and a water layer.
2 An experiment was. The reverse process when the vapor goes back to the solid phase is called deposition. This uses a solvent to selectively dissolve one component of the solid mixture.
With this technique a soluble solid can be separated from an insoluble solid. Lab 3 - Extraction Objective In this experiment you will separate the components of a commercial headache powder via an extractive process. This separation will be accomplished by taking advantage of the fact that each component contains different functional groups which will react differently when treated with a specific reagent.
Separation of a three-component mixture by extraction lab report help the compounds are benzoic acid 9-fluorene and ethyl-4- aminobenzoate. I need help to write Discussion about this lab report and I dont know what should I writeI got 25 recovary for benzoic acid 32 for ethyl-4- aminobenzoate and 50 recovary for 9-fluorene by the way I lost some. Lab Report 1 Two Base Extraction - Google Docs.
Two Base Extraction Jeremy Wolf. The purpose of this experiment is to use a two-base extraction method to separate a sample of three immiscible compounds. We converted both benzoic acid and 2-naphthol to their conjugate bases which are soluble.
Separation of a Three Component Mixture Background In Experiment 1 we introduced the idea of Like dissolves Like where a polar solvent water was able dissolve polar charged molecules but not neutral molecules that stayed in the organic layer. In this experiment you will start with 3 compounds carboxylic acid a basic amine and neutral all of which. 151-50 Fall 2013 Lab report Isolating the Components of a Three Component Mixture The purpose of this experiment was to separate the component of three mixtures sand sodium chloride and calcium carbonate then calculate the percentage by mass of each component recovered from the mixture.
A final report is prepared by the students in which they. Separation all support identity. Separation of a Three-Component Organic Mixture using Extraction Techniques and.
Pervaporation - dehydration of alcohols separation of azeotropic mixture etc. Be specific suggestions I should have heated the mixture to 550 C not general. Lab Instructor Date LAB REPORT EXPERIMENT 4 Extraction.
A Separation and Isolation Technique Separation of Three-Component Mixture Weight Compound. Of being mixed does not change the components of a mixture and they retain their individual. Chromatography is a technique used to separate components in a mixture and can be used to partially identify the components.
Blank lab report form mixture sample petri dish or other container such as a paper. Chromatography is a sophisticated method of separating and identifying mixtures of. Separation of a Three-Component Organic Mixture using Extraction Techniques and Acid-Base Chemistry Chemistry 233.
Organic Lab I August A. Gallo and Jimmy D. Fitzpatrick Department of Chemistry University of Southwestern Louisiana Lafayette LA 70504-4370 Revised 2003.